
8 Batch Processing Software Features You Must Have If You Use a Formula or Recipe for Manufacturing
Not all manufacturing is the same.
The fact is, most manufacturing software is built for discrete manufacturing. Even with customizations and add-on tools, this software is not the best fit for process and batch manufacturers.
Read: Compare Discrete Manufacturing versus Process and Batch Manufacturing – Key Differences 
Sadly, many manufacturers, and even the software consultants that advise them, think their needs are standard. As a result, they do not realize until it is too late that the software they implemented is missing critical pieces required to run their business.
In this post, I will outline eight features that batch manufacturers need in their software.
I will say upfront that all of these features are included in Vicinity Software. However, the information can be used to evaluate any manufacturing software on the market. I have used the example of a brewery, but the principles apply to companies across all industries that use a formula or recipe in their manufacturing process such as chemical or food manufacturers.
1. Ability to Change a Formula Based on Variability of Raw Materials
When we think about the variability of raw material lots, the ability to be able to change a formula based on a value in that lot is very important.
In Vicinity Sofware, when you are creating a batch, you can say, “Here’s the lot I’m going to use of this raw material. I need to add more or less of that particular ingredient, which may also affect some other ingredient.”
An example of this would be water content. You might add water to a batch, but if you have more percent water in a raw material lot, you can use less water additions. You are changing some ingredients of the standard formula to get to your desired end result.
A key feature of that is the modification of a batch based on the actual lot being used as a batch is being scaled.
2. Ability to Make the Same Product at Multiple Facilities
Many manufacturers make the same product at multiple locations. Sierra Nevada Brewing is a good example of this. They use a formula to produce their Pale Ale beer in the Mills River, NC facility. When they brew it in Chico, CA, they use a different formula because the water chemistry is different and they want it to taste exactly the same. They need to be able to set up a production facility, which is different from the site. The production location makes the same SKU but uses a different formula. The same finished good can have two different SKUs, one per facility, as an example.
Most discrete manufacturing software does not support making the same item using different BOMs. This is because batch manufacturers are more affected by variation or availability to raw materials. Vicinity Software ties a formula to a produced item by facility. This provides the needed flexibility for batch manufacturers.
3. Lot Traceability for Multiple End Items from One Batch
Batch manufacturers often make multiple end items from a single batch ticket. For example, manufacturers in the brewing industry can have the exact same beer in a keg, a can, or a bottle. That drives out some other challenges, like lot traceability. Some companies need to have the same lot number for all those products coming out. Other companies need those to be separate lot numbers. The software needs to be able to deal with that variability based on your specific needs. 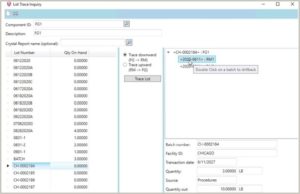
4. Quality Control for Multiple End Items from One Batch
When there are multiple end items on a batch ticket, you will have different quality control (QC) for each of those end items that are coming off the line. For example, fill level detection. We need to make sure that there is X amount of beer in the bottle. In addition to end item QC, there is also formula QC. Common QC needs to be done for the entire formula that’s going into each of those end items. You can’t just tie your QC to an SKU to a finished good. You also have to be able to tie it to the formula, which is the parent.
That simple concept of multiple finished goods from a batch starts having tentacles all through the system. In addition to lot traceability and quality control, scheduling is also impacted.
5. Scheduling for Multiple End Items from One Batch
Your manufacturing software needs to be able to schedule by formula, not just by SKU. If you are going to make a beer, you are typically scheduling the liquid, but you also need to be able to tie all those finished goods back to that same formula. Am I making the beer for cans, and then separately making the beer for kegs? No. You make one big, brite tank of beer and then you package from there. So that multiple end item against the batch ticket also affects scheduling, which gets into costing and some other related areas.
related areas.
6. Advanced Lot Traceability
Especially in the food world, 21 CFR part 11 is a big deal. Most people know it as lot traceability. It is an FDA requirement to be able to track your raw materials. Anything that goes into the product or touches the product has to be lot traced. That means everything except the box it is shipped in has to be lot tracked.
We have to track the intermediate raw material lot. You might have something in a holding tank for a while. Of course, the finished goods, and any packaging and the formula that goes into production has to be lot tracked and able to be tied from raw material to finished goods and back again. I think some discrete products do an okay job of that, but it becomes a bit more complicated for batch manufacturing because you get into rework, byproducts and things like that.
7. Variable Yield Analysis
One of the characteristics of batch manufacturing is variable yield. Each batch is a little bit different. One day you put in 100 pounds of raw materials and get 90 pounds out. The next day, you might get 85 pounds out. However, you will have to be able to predict the outcome for scheduling. For MRP, you need to be able to determine how much raw material you are going to buy and factor in the loss.
I think discrete manufacturing software does an okay job of loss at the SKU level. The challenge for batch manufacturing is that we get down to the line items, in other words, different ingredients.
When you scale up that batch and make a bigger batch, you may have a disproportionate amount of loss of one item versus another. For example, if I put water into a process or an ingredient has water in it, and it goes through an evaporation process, anything with water is going to have more loss than a solid. You have to be able to account for that when you scale up the formula.
Scaling is a lot more complicated, especially if you are using a lab formula and making a variable-size batch. This happens often in made-to-order operations. Each batch may be a different size. Additionally, it is common to scale a batch around the quantity of a raw material. You may want to use up an entire lot of a raw material and scale the batch around the quantity of a raw material lot.
Backflushing is a term used in discrete manufacturing that is not used in batch manufacturing. You need to be looking at actual input to actual output because your yields are different. If you don’t do that, you’ll get a flat yield, and you’ve lost any yield calculation, which affects your costs, which also affects scheduling MRP.
8. Industry-Specific Regulatory Compliance
Chemical or food manufacturers have different compliance issues than other manufacturers. 
Nutritional Labeling
Food manufacturers must do nutritional labeling. The information consumers recognize on the back panel on all food packaging is calculated from the formula. The information is coming from the ingredients you received from a supplier. The formula needs to be able to calculate that, give it to you as a percentage basis, and then also be able to take it out by serving sizes. Different products have different serving sizes. You need to be able to tell your software the serving size you are using and the nutritional calculations. Then a label goes on it, which consumers recognize. Of course, this is closely related to the lot traceability discussed earlier.
Hazardous Bill of Lading
Discrete manufacturers can normally get away with a straight bill of lading to show where it is going and what is in the box. Chemical manufacturers need to deal with hazardous bills of lading compliance. In the USA, there are certain DOT requirements and codes that need to put on each product. If there is an accident, the authorities can look at the bill of lading and know what steps need to be taken.
SDS-GHS (formerly MSDS)
Another compliance issue is SDS-GHS (formerly known as MSDS). About ten years ago authorities switched over from a US standard material safety data sheet to a more global system that is being used all across the world. Oddly enough, it was the United Nations that put that together. Now, there is consistency across the world with things like placards and visual pictograms. There is a lot of technical information that has to be disclosed in certain ways and that has to be created. It is all being read from your formula, which is read from the ingredients. And you guessed it, your software has to be able to handle this.
SARA 312/313
Finally, there are some very industry-specific compliance issues. I’ll just throw one out there. In the chemical world you have SARA 312/313. That basically is asking you to report, “How much hazardous stuff are you keeping at the plant, and how much have you used this year?” One side is kind of a balance sheet approach, what is your inventory? The other is more of an income statement question, what is the flow of how much have you used? Your software has to be able track and report all of that.
Compliance for batch manufacturing is a really big issue. Other manufacturing packages would need to be customized, which often leads to problems when there are changes and upgrades.
Next Steps
Of course, there are many ERP software packages available that have the word “manufacturing” in the title. The truth is that not all of them will work for the unique needs of the batch manufacturer.
Very likely, the firm that is selling you the ERP system does not even realize that something like the inability to do multiple facilities within Dynamics 365 Business Central will be an issue for a manufacturer. On the other hand, our team at Vicinity Software works with batch manufacturing requirements every day and understands the subtleties batch manufacturers face.
Vicinity Software does not sell ERP software. But we can work in partnership with your Microsoft Dynamics ERP provider to add on the specific manufacturing functionality that will make you successful.
If you use a recipe or formula in your manufacturing process, talk to us before you select any ERP software to make sure you get all the features you need for batch manufacturing.
Contact us online or call 770.421.2467.
By Randy Smith, CEO and Co-Founder, Vicinity Software, www.vicinitysoftware.com
About Vicinity Software:
Vicinity Software is the leading developer of process manufacturing solutions for the chemical (VicinityChem), food (VicinityFood) and brewing (VicinityBrew) markets. Established in 2001 and powered by a team of developers, implementation experts and business consultants-all US-based, Vicinity introduced the first software product written exclusively for formula or batch manufacturers who use QuickBooks Online® or Microsoft Dynamics® ERP solutions, including Dynamics GP, SL, NAV and BC.
If you use a formula or batch in your manufacturing process, we know that you are unique. Let’s start the conversation. Contact us today!
If you are not a customer of Vicinity Software, contact us today for a demo and to learn more about how we can support your business.

